 
Lewis structures don’t give us a 3-dimensional view of how the atoms are bonded together The Lewis structure implies a cross shape with 90o angles Would you have predicted this arrangement of atoms from just seeing it’s Lewis structure?ġ1 So how do we find the shape of a molecule?īy using the VSEPR Theory (pronounced Vess Purr)ġ2 Valence Shell Electron Pair Repulsion Theory The shape of a molecule may determine its properties and uses Properties such as smell, taste, and proper targeting (of drugs) are all possible because of the shapes of moleculesĨ Prostaglandin which causes inflammation (swelling) is produced by theĪspirin works because of its shape! Prostaglandin which causes inflammation (swelling) is produced by the COX-1 and COX-2 enzymes Aspirin can block the substrate from bonding to the COX-1 or COX-2 enzyme thus preventing the production of prostaglandinĩ The Complementary Shapes of an Enzyme and Its Substrate 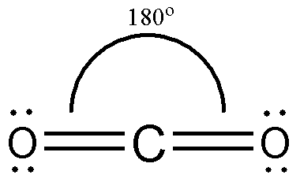
Molecular Geometry The SHAPES of moleculesħ Why the shape of a molecule is important Complete the following for each compound given: (1) Molecular Mass and Percent Composition (2a) Lewis Dot Structure and Electronic Geometry (Give Bonding & Nonbonding Domains) (3) Molecular Geometry or VSEPR Shapes (4) Fisher Projections Instructions In order to complete this probe, divide a sheet of lineless paper into quarters for each compound. 
Types of Bonds and Ionic/Covalent Compds due BEFORE Christmas Break All google classroom assignments due BEFORE Christmas Break The Organic Chemistry Lab due BEFORE Christmas Break Molecular Geometry Challenge due BEFORE Christmas Break Chemical Reactions Activity TBD H.C.3A.7 Analyze and interpret data to determine the empirical formula of a compound and the percent composition of a compound. Presentation on theme: "Get Molecular Geometry challenge sheet"- Presentation transcript:īR: Get Molecular Geometry challenge sheetĢ Standards H.C.3A.3 Analyze and interpret data to predict the type of bonding (ionic or covalent) and the shape of simple compounds by using the Lewis dot structures and oxidation numbers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
